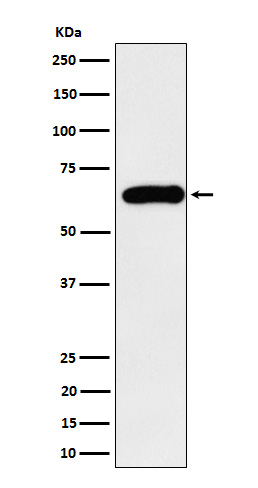
SQSTM1 / p62 Antibody
Rabbit mAb
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC, FC, ICC, IP |
|---|---|
| Primary Accession | Q13501 |
| Clonality | Monoclonal |
| Other Names | A170; DMRV; EBIAP; ORCA; OSF-6; Osi; OSIL; p60; p62; p62B; PDB3; Sequestosome 1; Sqstm1; STAP; STONE14; Ubiquitin binding protein p62; ZIP; ZIP3; |
| Isotype | Rabbit IgG |
| Host | Rabbit |
| Calculated MW | 47687 Da |
| Dilution | WB 1:500~1:2000 IHC 1:50~1:200 ICC/IF 1:50~1:200 IP 1:50 FC 1:50 |
|---|---|
| Purification | Affinity-chromatography |
| Immunogen | A synthesized peptide derived from human SQSTM1 / p62 |
| Description | Adapter protein which binds ubiquitin and may regulate the activation of NFKB1 by TNF-alpha, nerve growth factor (NGF) and interleukin-1. May play a role in titin/TTN downstream signaling in muscle cells. May regulate signaling cascades through ubiquitination. Adapter that mediates the interaction between TRAF6 and CYLD (By similarity). |
| Storage Condition and Buffer | Rabbit IgG in phosphate buffered saline , pH 7.4, 150mM NaCl, 0.02% sodium azide and 50% glycerol. Store at +4°C short term. Store at -20°C long term. Avoid freeze / thaw cycle. |
| Name | SQSTM1 {ECO:0000303|PubMed:16286508, ECO:0000312|HGNC:HGNC:11280} |
|---|---|
| Function | Molecular adapter required for selective macroautophagy (aggrephagy) by acting as a bridge between polyubiquitinated proteins and autophagosomes (PubMed:15340068, PubMed:15953362, PubMed:16286508, PubMed:17580304, PubMed:20168092, PubMed:22017874, PubMed:22622177, PubMed:24128730, PubMed:28404643, PubMed:29343546, PubMed:29507397, PubMed:31857589, PubMed:33509017, PubMed:34471133, PubMed:34893540, PubMed:35831301, PubMed:37306101, PubMed:37802024). Promotes the recruitment of ubiquitinated cargo proteins to autophagosomes via multiple domains that bridge proteins and organelles in different steps (PubMed:16286508, PubMed:20168092, PubMed:22622177, PubMed:24128730, PubMed:28404643, PubMed:29343546, PubMed:29507397, PubMed:34893540, PubMed:37802024). SQSTM1 first mediates the assembly and removal of ubiquitinated proteins by undergoing liquid-liquid phase separation upon binding to ubiquitinated proteins via its UBA domain, leading to the formation of insoluble cytoplasmic inclusions, known as p62 bodies (PubMed:15911346, PubMed:20168092, PubMed:22017874, PubMed:24128730, PubMed:29343546, PubMed:29507397, PubMed:31857589, PubMed:37802024). SQSTM1 then interacts with ATG8 family proteins on autophagosomes via its LIR motif, leading to p62 body recruitment to autophagosomes, followed by autophagic clearance of ubiquitinated proteins (PubMed:16286508, PubMed:17580304, PubMed:20168092, PubMed:22622177, PubMed:24128730, PubMed:28404643, PubMed:37802024). SQSTM1 is itself degraded along with its ubiquitinated cargos (PubMed:16286508, PubMed:17580304, PubMed:37802024). Also required to recruit ubiquitinated proteins to PML bodies in the nucleus (PubMed:20168092). Also involved in autophagy of peroxisomes (pexophagy) in response to reactive oxygen species (ROS) by acting as a bridge between ubiquitinated PEX5 receptor and autophagosomes (PubMed:26344566). Acts as an activator of the NFE2L2/NRF2 pathway via interaction with KEAP1: interaction inactivates the BCR(KEAP1) complex by sequestering the complex in inclusion bodies, promoting nuclear accumulation of NFE2L2/NRF2 and subsequent expression of cytoprotective genes (PubMed:20452972, PubMed:28380357, PubMed:33393215, PubMed:37306101). Promotes relocalization of 'Lys-63'-linked ubiquitinated STING1 to autophagosomes (PubMed:29496741). Involved in endosome organization by retaining vesicles in the perinuclear cloud: following ubiquitination by RNF26, attracts specific vesicle-associated adapters, forming a molecular bridge that restrains cognate vesicles in the perinuclear region and organizes the endosomal pathway for efficient cargo transport (PubMed:27368102, PubMed:33472082). Sequesters tensin TNS2 into cytoplasmic puncta, promoting TNS2 ubiquitination and proteasomal degradation (PubMed:25101860). May regulate the activation of NFKB1 by TNF-alpha, nerve growth factor (NGF) and interleukin-1 (PubMed:10356400, PubMed:10747026, PubMed:11244088, PubMed:12471037, PubMed:16079148, PubMed:19931284). May play a role in titin/TTN downstream signaling in muscle cells (PubMed:15802564). Adapter that mediates the interaction between TRAF6 and CYLD (By similarity). |
| Cellular Location | Cytoplasmic vesicle, autophagosome. Preautophagosomal structure. Cytoplasm, cytosol. Nucleus, PML body. Late endosome. Lysosome. Nucleus Endoplasmic reticulum. Cytoplasm, myofibril, sarcomere {ECO:0000250|UniProtKB:O08623}. Note=In cardiac muscle, localizes to the sarcomeric band (By similarity). Localizes to cytoplasmic membraneless inclusion bodies, known as p62 bodies, containing polyubiquitinated protein aggregates (PubMed:11786419, PubMed:20357094, PubMed:22017874, PubMed:29343546, PubMed:29507397, PubMed:31857589, PubMed:37306101, PubMed:37802024). In neurodegenerative diseases, detected in Lewy bodies in Parkinson disease, neurofibrillary tangles in Alzheimer disease, and HTT aggregates in Huntington disease (PubMed:15158159). In protein aggregate diseases of the liver, found in large amounts in Mallory bodies of alcoholic and nonalcoholic steatohepatitis, hyaline bodies in hepatocellular carcinoma, and in SERPINA1 aggregates (PubMed:11981755) Enriched in Rosenthal fibers of pilocytic astrocytoma (PubMed:11786419). In the cytoplasm, observed in both membrane-free ubiquitin-containing protein aggregates (sequestosomes) and membrane- surrounded autophagosomes (PubMed:15953362, PubMed:17580304) Colocalizes with TRIM13 in the perinuclear endoplasmic reticulum (PubMed:22178386). Co-localizes with TRIM5 in cytoplasmic bodies (PubMed:20357094). When nuclear export is blocked by treatment with leptomycin B, accumulates in PML bodies (PubMed:20168092) {ECO:0000250|UniProtKB:O08623, ECO:0000269|PubMed:11786419, ECO:0000269|PubMed:11981755, ECO:0000269|PubMed:15158159, ECO:0000269|PubMed:15953362, ECO:0000269|PubMed:17580304, ECO:0000269|PubMed:20168092, ECO:0000269|PubMed:20357094, ECO:0000269|PubMed:22017874, ECO:0000269|PubMed:22178386, ECO:0000269|PubMed:29343546, ECO:0000269|PubMed:29507397, ECO:0000269|PubMed:31857589, ECO:0000269|PubMed:37306101, ECO:0000269|PubMed:37802024} |
| Tissue Location | Ubiquitously expressed. |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.











 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.