NBN Antibody (C-term)
Affinity Purified Rabbit Polyclonal Antibody (Pab)
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, FC, IF, E |
|---|---|
| Primary Accession | O60934 |
| Other Accession | NP_002476.2 |
| Reactivity | Human |
| Host | Rabbit |
| Clonality | Polyclonal |
| Isotype | Rabbit IgG |
| Calculated MW | 84959 Da |
| Antigen Region | 602-630 aa |
| Gene ID | 4683 |
|---|---|
| Other Names | Nibrin, Cell cycle regulatory protein p95, Nijmegen breakage syndrome protein 1, NBN, NBS, NBS1, P95 |
| Target/Specificity | This NBN antibody is generated from rabbits immunized with a KLH conjugated synthetic peptide between 602-630 amino acids from the C-terminal region of human NBN. |
| Dilution | IF~~1:10~50 WB~~1:1000 FC~~1:10~50 |
| Format | Purified polyclonal antibody supplied in PBS with 0.09% (W/V) sodium azide. This antibody is purified through a protein A column, followed by peptide affinity purification. |
| Storage | Maintain refrigerated at 2-8°C for up to 2 weeks. For long term storage store at -20°C in small aliquots to prevent freeze-thaw cycles. |
| Precautions | NBN Antibody (C-term) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | NBN (HGNC:7652) |
|---|---|
| Function | Component of the MRN complex, which plays a central role in double-strand break (DSB) repair, DNA recombination, maintenance of telomere integrity and meiosis (PubMed:10888888, PubMed:15616588, PubMed:18411307, PubMed:18583988, PubMed:18678890, PubMed:19759395, PubMed:23115235, PubMed:28216226, PubMed:28867292, PubMed:9705271). The MRN complex is involved in the repair of DNA double-strand breaks (DSBs) via homologous recombination (HR), an error-free mechanism which primarily occurs during S and G2 phases (PubMed:19759395, PubMed:28867292, PubMed:9705271). The complex (1) mediates the end resection of damaged DNA, which generates proper single-stranded DNA, a key initial steps in HR, and is (2) required for the recruitment of other repair factors and efficient activation of ATM and ATR upon DNA damage (PubMed:19759395, PubMed:9705271). The MRN complex possesses single-strand endonuclease activity and double-strand-specific 3'-5' exonuclease activity, which are provided by MRE11, to initiate end resection, which is required for single-strand invasion and recombination (PubMed:19759395, PubMed:28867292, PubMed:9705271). Within the MRN complex, NBN acts as a protein-protein adapter, which specifically recognizes and binds phosphorylated proteins, promoting their recruitment to DNA damage sites (PubMed:12419185, PubMed:15616588, PubMed:18411307, PubMed:18582474, PubMed:18583988, PubMed:18678890, PubMed:19759395, PubMed:19804756, PubMed:23762398, PubMed:24534091, PubMed:27814491, PubMed:27889449, PubMed:33836577). Recruits MRE11 and RAD50 components of the MRN complex to DSBs in response to DNA damage (PubMed:12419185, PubMed:18411307, PubMed:18583988, PubMed:18678890, PubMed:24534091, PubMed:26438602). Promotes the recruitment of PI3/PI4-kinase family members ATM, ATR, and probably DNA-PKcs to the DNA damage sites, activating their functions (PubMed:15064416, PubMed:15616588, PubMed:15790808, PubMed:16622404, PubMed:22464731, PubMed:30952868, PubMed:35076389). Mediates the recruitment of phosphorylated RBBP8/CtIP to DSBs, leading to cooperation between the MRN complex and RBBP8/CtIP to initiate end resection (PubMed:19759395, PubMed:27814491, PubMed:27889449, PubMed:33836577). RBBP8/CtIP specifically promotes the endonuclease activity of the MRN complex to clear DNA ends containing protein adducts (PubMed:27814491, PubMed:27889449, PubMed:30787182, PubMed:33836577). The MRN complex is also required for the processing of R-loops (PubMed:31537797). NBN also functions in telomere length maintenance via its interaction with TERF2: interaction with TERF2 during G1 phase preventing recruitment of DCLRE1B/Apollo to telomeres (PubMed:10888888, PubMed:28216226). NBN also promotes DNA repair choice at dysfunctional telomeres: NBN phosphorylation by CK2 promotes non- homologous end joining repair at telomeres, while unphosphorylated NBN promotes microhomology-mediated end-joining (MMEJ) repair (PubMed:28216226). Enhances AKT1 phosphorylation possibly by association with the mTORC2 complex (PubMed:23762398). |
| Cellular Location | Nucleus. Chromosome. Nucleus, PML body. Chromosome, telomere Note=Localizes to discrete nuclear foci after treatment with genotoxic agents (PubMed:10783165, PubMed:26215093, PubMed:26438602). Localizes to DNA double-strand breaks (DSBs); recruited to DNA damage sites via association with phosphorylated proteins, such as phosphorylated H2AX, phosphorylated MDC1 and phosphorylated RAD17 (PubMed:12419185, PubMed:18411307, PubMed:18582474, PubMed:18583988, PubMed:18678890, PubMed:19338747, PubMed:23115235, PubMed:24534091, PubMed:26438602) Acetylation of 'Lys-5' of histone H2AX (H2AXK5ac) promotes NBN/NBS1 assembly at the sites of DNA damage (PubMed:26438602) |
| Tissue Location | Ubiquitous (PubMed:9590180). Expressed at high levels in testis (PubMed:9590180). |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
Mutations in this gene are associated with Nijmegen breakage syndrome, an autosomal recessive chromosomal instability syndrome characterized by microcephaly, growth retardation, immunodeficiency, and cancer predisposition. The encoded protein is a member of the MRE11/RAD50 double-strand break repair complex which consists of 5 proteins. This gene product is thought to be involved in DNA double-strand break repair and DNA damage-induced checkpoint activation.
References
Liu, Y., et al. Carcinogenesis 31(10):1762-1769(2010)
Kavitha, C.V., et al. Biochem. Biophys. Res. Commun. 399(4):575-580(2010)
Ho-Pun-Cheung, A., et al. Pharmacogenomics J. (2010) In press :
Jelonek, K., et al. J. Appl. Genet. 51(3):343-352(2010)
Jugessur, A., et al. PLoS ONE 5 (7), E11493 (2010) :
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.




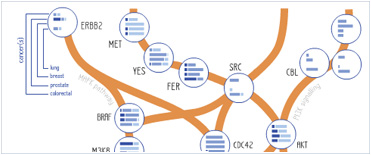







 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.




