RPA3 Antibody (clone 111)
Mouse Monoclonal Antibody
- SPECIFICATION
- CITATIONS
- PROTOCOLS
- BACKGROUND

Application
| WB, IHC-P, IP |
|---|---|
| Primary Accession | P35244 |
| Reactivity | Human, Mouse |
| Host | Mouse |
| Clonality | Monoclonal |
| Clone Names | 111 |
| Calculated MW | 14kDa |
| Dilution | IHC-P (5 µg/ml) |
| Gene ID | 6119 |
|---|---|
| Other Names | Replication protein A 14 kDa subunit, RP-A p14, Replication factor A protein 3, RF-A protein 3, RPA3, REPA3, RPA14 |
| Target/Specificity | Full-length human RPA-14 expressed in E. coli. |
| Reconstitution & Storage | Long term: -20°C; Short term: +4°C. Avoid repeat freeze-thaw cycles. |
| Precautions | RPA3 Antibody (clone 111) is for research use only and not for use in diagnostic or therapeutic procedures. |
| Name | RPA3 |
|---|---|
| Synonyms | REPA3, RPA14 |
| Function | As part of the heterotrimeric replication protein A complex (RPA/RP-A), binds and stabilizes single-stranded DNA intermediates that form during DNA replication or upon DNA stress. It prevents their reannealing and in parallel, recruits and activates different proteins and complexes involved in DNA metabolism. Thereby, it plays an essential role both in DNA replication and the cellular response to DNA damage (PubMed:17596542, PubMed:9430682). In the cellular response to DNA damage, the RPA complex controls DNA repair and DNA damage checkpoint activation. Through recruitment of ATRIP activates the ATR kinase a master regulator of the DNA damage response (PubMed:24332808). It is required for the recruitment of the DNA double-strand break repair factors RAD51 and RAD52 to chromatin, in response to DNA damage. Also recruits to sites of DNA damage proteins like XPA and XPG that are involved in nucleotide excision repair and is required for this mechanism of DNA repair (PubMed:7697716). Also plays a role in base excision repair (BER), probably through interaction with UNG (PubMed:9765279). RPA stimulates 5'-3' helicase activity of BRIP1/FANCJ (PubMed:17596542). Also recruits SMARCAL1/HARP, which is involved in replication fork restart, to sites of DNA damage. May also play a role in telomere maintenance. RPA3 has its own single-stranded DNA-binding activity and may be responsible for polarity of the binding of the complex to DNA (PubMed:19010961). As part of the alternative replication protein A complex, aRPA, binds single-stranded DNA and probably plays a role in DNA repair. Compared to the RPA2-containing, canonical RPA complex, may not support chromosomal DNA replication and cell cycle progression through S-phase. The aRPA may not promote efficient priming by DNA polymerase alpha but could support DNA synthesis by polymerase delta in presence of PCNA and replication factor C (RFC), the dual incision/excision reaction of nucleotide excision repair and RAD51-dependent strand exchange (PubMed:19996105). |
| Cellular Location | Nucleus |

Thousands of laboratories across the world have published research that depended on the performance of antibodies from Abcepta to advance their research. Check out links to articles that cite our products in major peer-reviewed journals, organized by research category.
info@abcepta.com, and receive a free "I Love Antibodies" mug.
Provided below are standard protocols that you may find useful for product applications.
Background
As part of the heterotrimeric replication protein A complex (RPA/RP-A), binds and stabilizes single-stranded DNA intermediates that form during DNA replication or upon DNA stress. It prevents their reannealing and in parallel, recruits and activates different proteins and complexes involved in DNA metabolism. Thereby, it plays an essential role both in DNA replication and the cellular response to DNA damage (PubMed:9430682). In the cellular response to DNA damage, the RPA complex controls DNA repair and DNA damage checkpoint activation. Through recruitment of ATRIP activates the ATR kinase a master regulator of the DNA damage response (PubMed:24332808). It is required for the recruitment of the DNA double-strand break repair factors RAD51 and RAD52 to chromatin, in response to DNA damage. Also recruits to sites of DNA damage proteins like XPA and XPG that are involved in nucleotide excision repair and is required for this mechanism of DNA repair (PubMed:7697716). Plays also a role in base excision repair (BER), probably through interaction with UNG (PubMed:9765279). Through RFWD3 may activate CHEK1 and play a role in replication checkpoint control. Also recruits SMARCAL1/HARP, which is involved in replication fork restart, to sites of DNA damage. May also play a role in telomere maintenance. RPA3 has its own single-stranded DNA-binding activity and may be responsible for polarity of the binding of the complex to DNA (PubMed:19010961). As part of the alternative replication protein A complex, aRPA, binds single-stranded DNA and probably plays a role in DNA repair. Compared to the RPA2-containing, canonical RPA complex, may not support chromosomal DNA replication and cell cycle progression through S-phase. The aRPA may not promote efficient priming by DNA polymerase alpha but could support DNA synthesis by polymerase delta in presence of PCNA and replication factor C (RFC), the dual incision/excision reaction of nucleotide excision repair and RAD51-dependent strand exchange (PubMed:19996105).
References
Umbricht C.B.,et al.J. Biol. Chem. 268:6131-6138(1993).
Kalnine N.,et al.Submitted (MAY-2003) to the EMBL/GenBank/DDBJ databases.
Hillier L.W.,et al.Nature 424:157-164(2003).
Aboussekhra A.,et al.Cell 80:859-868(1995).
Keshav K.F.,et al.Mol. Cell. Biol. 15:3119-3128(1995).
If you have used an Abcepta product and would like to share how it has performed, please click on the "Submit Review" button and provide the requested information. Our staff will examine and post your review and contact you if needed.
If you have any additional inquiries please email technical services at tech@abcepta.com.




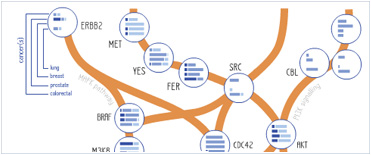







 Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them.
Foundational characteristics of cancer include proliferation, angiogenesis, migration, evasion of apoptosis, and cellular immortality. Find key markers for these cellular processes and antibodies to detect them. The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle.
The SUMOplot™ Analysis Program predicts and scores sumoylation sites in your protein. SUMOylation is a post-translational modification involved in various cellular processes, such as nuclear-cytosolic transport, transcriptional regulation, apoptosis, protein stability, response to stress, and progression through the cell cycle. The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.
The Autophagy Receptor Motif Plotter predicts and scores autophagy receptor binding sites in your protein. Identifying proteins connected to this pathway is critical to understanding the role of autophagy in physiological as well as pathological processes such as development, differentiation, neurodegenerative diseases, stress, infection, and cancer.


